Write notes on 1) Electron affinity 2) Electronegativity 3) Magnetic properties of elements.
Introduction
Electron affinity and electronegativity are fundamental concepts in chemistry that describe an atom's ability to attract and retain electrons. These properties are crucial for understanding chemical bonding, molecular structure, and reactivity. For students preparing for the Collegeboard AP Chemistry exam, grasping these concepts is essential for mastering periodic trends and predicting the behavior of elements.
Key Concepts
Electron Affinity: Definition and Significance
Electron affinity refers to the amount of energy released when an isolated gaseous atom gains an electron to form a negative ion. It is a measure of an atom's ability to accept an additional electron. Electron affinity plays a significant role in chemical reactions, particularly in the formation of anions and the stability of molecules.
The general reaction for electron affinity can be represented as:
A higher electron affinity indicates a greater tendency to accept electrons, which is a vital factor in the reactivity of nonmetals. Most elements have a positive electron affinity, meaning they release energy when gaining an electron. However, some noble gases have little to no electron affinity due to their complete valence electron shells.
Electronegativity: Definition and Significance
Electronegativity is a measure of an atom's ability to attract and hold onto electrons within a chemical bond. Unlike electron affinity, which pertains to the addition of an electron to an isolated atom, electronegativity describes the behavior of an atom in a molecule. It is a dimensionless quantity and is typically measured on the Pauling scale.
Electronegativity influences the polarity of bonds, molecular geometry, and the overall reactivity of compounds. Atoms with high electronegativity tend to attract electrons more strongly, leading to polar covalent bonds, while those with low electronegativity may donate electrons, resulting in nonpolar or ionic bonds.
Periodic Trends of Electron Affinity and Electronegativity
Both electron affinity and electronegativity exhibit distinct trends across the periodic table:
- Across a Period: Electron affinity generally becomes more negative from left to right due to increasing nuclear charge, which enhances the atom's ability to attract electrons. Similarly, electronegativity increases across a period as atoms more effectively draw bonding electrons.
- Down a Group: Electron affinity becomes less negative down a group because the added electron is placed farther from the nucleus, reducing the attraction. Electronegativity also decreases down a group for the same reason, as the outer electrons are less strongly attracted to the nucleus.
Exceptions to these trends can occur due to electron configurations and subshell electron repulsions. For example, the electron affinity of nitrogen is less negative than that of carbon, despite its position to the right, because nitrogen's half-filled p-orbital provides extra stability.
Relationship Between Electron Affinity and Electronegativity
While electron affinity and electronegativity are related, they are not identical. Electron affinity is an intrinsic property of an isolated atom, whereas electronegativity is a comparative measure between atoms in a bond. High electron affinity often correlates with high electronegativity, as both indicate a strong ability to attract electrons. However, factors like atomic radius and bonding environment can influence their values independently.
Measuring and Comparing Electron Affinity and Electronegativity
Electron affinity is measured in kilojoules per mole (kJ/mol) and can be experimentally determined using techniques like photoelectron spectroscopy. Electronegativity, being a dimensionless value, is derived from various scales, with the Pauling scale being the most widely used. Other scales include the Mulliken and Allred-Rochow scales, each considering different aspects of atomic properties.
Understanding these measurements allows chemists to predict and explain the behavior of elements in chemical reactions and bonding scenarios.
Applications of Electron Affinity and Electronegativity
These properties are essential in several areas of chemistry:
- Covalent Bonding: Electronegativity differences between atoms determine the type of covalent bond—nonpolar, polar, or ionic.
- Redox Reactions: High electron affinity elements are strong oxidizing agents as they readily gain electrons.
- Material Science: Designing materials with specific electronic properties relies on understanding electron affinity and electronegativity.
- Biochemistry: Enzyme function and molecular interactions are influenced by the electronegativity of constituent atoms.
These applications highlight the importance of electron affinity and electronegativity in both theoretical and practical chemistry.
Challenges in Understanding Electron Affinity and Electronegativity
Despite their importance, students often face challenges in mastering these concepts:
- Conceptual Overlap: Distinguishing between electron affinity and electronegativity can be confusing due to their related nature.
- Periodic Trends Exceptions: Anomalies in periodic trends require a deeper understanding of electron configurations and atomic structure.
- Quantitative Comparisons: Applying numerical values of electron affinity and electronegativity in problem-solving demands precision and familiarity with different scales.
Overcoming these challenges involves focused study on periodic trends, practice with various examples, and a solid grasp of underlying atomic principles.
Comparison Table
| Aspect | Electron Affinity | Electronegativity |
|---|---|---|
| Definition | Energy change when an atom gains an electron | Ability of an atom to attract electrons in a bond |
| Measurement Units | kJ/mol | Dimensionless (Pauling scale) |
| Periodic Trend Across a Period | Generally more negative | Increases |
| Periodic Trend Down a Group | Less negative | Decreases |
| Relationship | Intrinsic property of an isolated atom | Comparative property between bonded atoms |
| Applications | Redox reactions, ion formation | Covalent bonding, molecular polarity |
| Typical Elements with High Values | Halogens | Fluorine, Oxygen |
Summary and Key Takeaways
- Electron affinity measures the energy change when an atom gains an electron, indicating its tendency to form negative ions.
- Electronegativity assesses an atom's ability to attract electrons within a chemical bond, influencing bond polarity.
- Both properties exhibit periodic trends, increasing across periods and decreasing down groups, with notable exceptions.
- Understanding these concepts is essential for predicting chemical behavior, bond formation, and reactivity.
- Mastery of electron affinity and electronegativity is crucial for success in the Collegeboard AP Chemistry exam.
The magnetic moment of a system measures the strength and the direction of its magnetism. The term itself usually refers to the magnetic dipole moment. Anything that is magnetic, like a bar magnet or a loop of electric current, has a magnetic moment. A magnetic moment is a vector quantity, with a magnitude and a direction. An electron has an electron magnetic dipole moment, generated by the electron's intrinsic spin property, making it an electric charge in motion. There are many different magnetic behavior including paramagnetism, diamagnetism, and ferromagnetism.
An interesting characteristic of transition metals is their ability to form magnets. Metal complexes that have unpaired electrons are magnetic. Since the last electrons reside in the d orbitals, this magnetism must be due to having unpaired d electrons. The spin of a single electron is denoted by the quantum number as +(1/2) or –(1/2). This spin is negated when the electron is paired with another, but creates a weak magnetic field when the electron is unpaired. More unpaired electrons increase the paramagnetic effects. The electron configuration of a transition metal (d-block) changes in a coordination compound; this is due to the repulsive forces between electrons in the ligands and electrons in the compound. Depending on the strength of the ligand, the compound may be paramagnetic or diamagnetic.
Ferromagnetism (Permanent Magnet)
Ferromagnetism is the basic mechanism by which certain materials (such as iron) form permanent magnets. This means the compound shows permanent magnetic properties rather than exhibiting them only in the presence of an external magnetic field (Figure ). In a ferromagnetic element, electrons of atoms are grouped into domains in which each domain has the same charge. In the presence of a magnetic field, these domains line up so that charges are parallel throughout the entire compound. Whether a compound can be ferromagnetic or not depends on its number of unpaired electrons and on its atomic size.
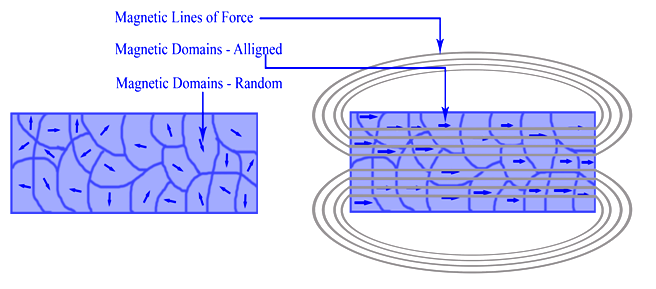
Figure : Ferromagnetism (a) nonmagnatized material and (2) Magnetized material with corresponding magnetic fields shown. Ferromagnetism, the permanent magnetism associated with nickel, cobalt, and iron, is a common occurrence in everyday life. Examples of the knowledge and application of ferromagnetism include Aristotle's discussion in 625 BC, the use of the compass in 1187, and the modern-day refrigerator. Einstein demonstrated that electricity and magnetism are inextricably linked in his theory of special relativity.
Paramagnetism (Attracted to Magnetic Field)
Paramagnetism refers to the magnetic state of an atom with one or more unpaired electrons. The unpaired electrons are attracted by a magnetic field due to the electrons' magnetic dipole moments. Hund's Rule states that electrons must occupy every orbital singly before any orbital is doubly occupied. This may leave the atom with many unpaired electrons. Because unpaired electrons can spin in either direction, they display magnetic moments in any direction. This capability allows paramagnetic atoms to be attracted to magnetic fields. Diatomic oxygen, is a good example of paramagnetism (described via molecular orbital theory). The following video shows liquid oxygen attracted into a magnetic field created by a strong magnet:
Video : Paramagnetism of Liquid Oxygen Diamagnetism (Repelled by Magnetic Field)
As shown in the video, molecular oxygen () is paramagnetic and is attracted to the magnet. In contrast, molecular nitrogen () has no unpaired electrons and is diamagnetic; it is unaffected by the magnet. Diamagnetic substances are characterized by paired electrons, e.g., no unpaired electrons. According to the Pauli Exclusion Principle which states that no two electrons may occupy the same quantum state at the same time, the electron spins are oriented in opposite directions. This causes the magnetic fields of the electrons to cancel out; thus there is no net magnetic moment, and the atom cannot be attracted into a magnetic field. In fact, diamagnetic substances are weakly repelled by a magnetic field as demonstrated with the pyrolytic carbon sheet in Figure .

Figure : Levitating pyrolytic carbon: A small (~6 mm) piece of pyrolytic graphite levitating over a permanent neodymium magnet array (5 mm cubes on a piece of steel). Note that the poles of the magnets are aligned vertically and alternate (two with north facing up, and two with south facing up, diagonally). (Public Domain; Splarka via Wikipedia). The magnetic properties of a substance can be determined by examining its electron configuration: If it has unpaired electrons, then the substance is paramagnetic and if all electrons are paired, the substance is then diamagnetic. This process can be broken into three steps:
- Write down the electron configuration
- Draw the valence orbitals
- Identify if unpaired electrons exist
- Determine whether the substance is paramagnetic or diamagnetic
Step 1: Find the electron configuration
For Cl atoms, the electron configuration is 3s23p5
Step 2: Draw the valence orbitals
Ignore the core electrons and focus on the valence electrons only.
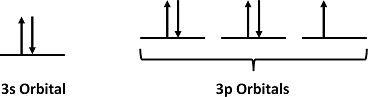
Step 3: Look for unpaired electrons
There is one unpaired electron.
Step 4: Determine whether the substance is paramagnetic or diamagnetic
Since there is an unpaired electron, atoms are paramagnetic (albeit, weakly).
Step 1: Find the electron configuration
For Zn atoms, the electron configuration is 4s23d10
Step 2: Draw the valence orbitals
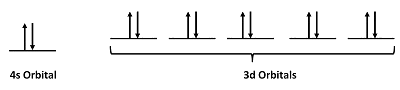
Step 3: Look for unpaired electrons
There are no unpaired electrons.
Step 4: Determine whether the substance is paramagnetic or diamagnetic
Because there are no unpaired electrons, atoms are diamagnetic.
Comments
Post a Comment